Alternative Medicines and Therapies
Alternatives to conventional medical care are increasingly popular in the United States, and their growing use by consumers represents a major trend in Western medicine. Alternative therapies appear to be used most frequently for medical conditions that are chronic , such as back pain, arthritis , sleep disorders, headache, and digestive problems. Surveys of U.S. consumers have shown that more people visit alternative practitioners each year than visit conventional primary-care physicians. Consumers do not necessarily reject conventional medicine, however. Many simply feel that alternative modalities offer complementary approaches that are more in line with their personal health philosophies.
Alternative Medicine, Complementary Medicine, and Integrative Medicine
The terms alternative medicine and alternative therapies refer to those medical practices that are not considered to be conventional medicine, as practiced in the United States. Other cultures, however, may use one or more of these approaches regularly, and, in fact, many have done so for thousands of years. Most people in the United States who use alternative medicine do so to complement conventional approaches. For example, in addition to using anti-inflammatory drugs to ease muscle pain, they may also use massage, chiropractic , and/or osteopathic manipulation. This practice of complementing conventional medicine with alternative approaches has given rise to the term complementary medicine. Presently, alternative medicine is most commonly referred to as complementary and alternative medicine (CAM). As conventional medical practitioners become familiar with alternative approaches, these approaches are being integrated into conventional medicine, which is giving rise to integrative medicine, in which a combination of therapies representing the best of conventional and alternative medicine is used.
Types of CAM Modalities
The National Center for Complementary and Alternative Medicine divides the various CAM modalities into five categories: (1) alternative medical systems, (2) mind-body interventions, (3) biologically-based treatments, (4) manipulative and body-based methods, and (5) energy therapies. These modalities include a wide variety of approaches, from acupuncture to nutrition to meditation to chiropractic.
Alternative medical systems include medical practices that are traditional in other cultures, such as the ayurvedic medical system of India, Chinese traditional medicine, and traditional Native American and Hawaiian medicine.
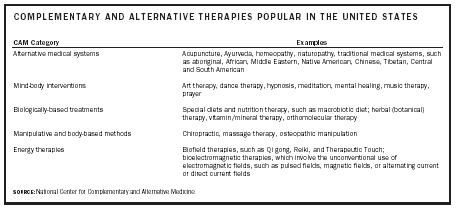
| CAM Category | Examples |
| SOURCE: National Center for Complementary and Alternative Medicine | |
| Alternative medical systems | Acupuncture, Ayurveda, homeopathy, naturopathy, traditional medical systems, such as aboriginal, African, Middle Eastern, Native American, Chinese, Tibetan, Central and South American |
| Mind-body interventions | Art therapy, dance therapy, hypnosis, meditation, mental healing, music therapy, prayer |
| Biologically-based treatments | Special diets and nutrition therapy, such as macrobiotic diet; herbal (botanical) therapy, vitamin/mineral therapy, orthomolecular therapy |
| Manipulative and body-based methods | Chiropractic, massage therapy, osteopathic manipulation |
| Energy therapies | Biofield therapies, such as Qi gong, Reiki, and Therapeutic Touch; bioelectromagnetic therapies, which involve the unconventional use of electromagnetic fields, such as pulsed fields, magnetic fields, or alternating current or direct current fields |
Mind-body interventions recognize the connection between the physical body and the spiritual self, and include practices such as meditation, prayer, and music therapy. Biologically-based modalities are primarily nutrition-related and vary from special diets such as the macrobiotic diet to the inclusion of dietary supplements in the diet. Body-based methods involve hands-on manipulation of the body, and include such modalities as massage and chiropractic. The energy therapies are based on the concept that the body has an energy field that can be manipulated to promote healing.
Included among the nutrition approaches that make up the biologicallybased modalities is the use of dietary supplements. Dietary supplements may be botanical ( herbal ) supplements or nutritional supplements, which include vitamins , minerals , antioxidants , enzymes , metabolites , nonprescription hormones , glandular extracts, and various amino acids , fatty acids , and other nutrients .
The Dietary Supplement Health and Education Act of 1994
Dietary supplement usage in the United States has increased significantly since the passage in 1994 of the Dietary Supplement Health and Education Act (DSHEA, pronounced Dee-shay). This legislation defined dietary supplements as distinct from food and drugs, and it allowed them to be sold without a prescription. The passage of DSHEA provided consumers with the right to purchase dietary supplements that they felt would help them attain their personal health goals. At the same time, DSHEA transferred to consumers the responsibility for making informed choices about the supplements that they used. In contrast to prescription and over-thecounter drugs, where effectiveness and safety must be demonstrated prior to marketing of the drugs, premarket approval is not required of manufacturers of dietary supplements. As a result, there is a greater potential risk that dietary supplements may be ineffective, or even harmful, as compared with drugs.
The dietary supplements industry is not unregulated, it is just not regulated to the extent that U.S. consumers have come to expect for prescription
![The rising popularity of alternative medicine has revived ancient techniques such as acupuncture. In the United States, the requirements for acupuncture licensure may vary from state to state. [Photograph by Yoav Levy. Phototake NYC. Reproduced by permission.]](../images/nwaz_01_img0022.jpg)
Supplement Facts Label
To help consumers make informed choices, dietary supplements now contain a supplement facts panel that clearly labels the product as a dietary supplement and gives information such as the amount of a standard dose, the number of recommended doses per day, the list of components (and how much of each is present in a standard dose), and, if the product is a botanical, the Latin name of the plant and the part of the plant used to prepare the product. This latter information is important because the components responsible for a certain health effect may be in one part of the plant but not in other parts. For example, a consumer purchasing ginger to protect against seasickness would want a product prepared from the roots of the plant, where the active components are, and not the leaves.
In addition, DSHEA established standards for terms, such as high potency and antioxidant, and for the types of claims that could be made for a product. Claims that a dietary supplement may help to prevent or cure a disease cannot be made. Such claims are considered health claims and must be reviewed by the Food and Drug Administration for scientific accuracy prior to approval for use on a product label. Manufacturers may, however, use structure/function claims that state that a product can, for example, "help to promote healthy blood levels of cholesterol ," but they may not state that a product "helps to prevent heart disease ." All structure/function claims must be accompanied by the following disclaimer: "This statement has not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease."
Selecting a CAM Modality
When selecting a CAM modality, it is important to have clearly defined health goals. In other words, what are you trying to accomplish, and is this modality an appropriate fit for you? If selecting a therapy, determine if the practitioner of the therapy being considered is a licensed health care practitioner. Licensure does not guarantee the modality will successfully meet one's needs, but it does provide some assurance of training and competency on the part of the practitioner. This information can usually be obtained from the various state boards of medicine, which are responsible for licensing health care professionals.
Selecting quality dietary supplements can be a bit more challenging. Both the natural products industry and the Food and Drug Administration are working to develop uniform standards of quality for dietary supplements. Until these standards are in place, however, consumers must be proactive in determining for themselves what supplements are consistent with their health goals and what manufacturers offer quality products. It is important not to be fooled by hype . Be wary of supplements that sound too good to be true or that promise to cure a medical condition.
Quality natural ingredients and responsible product testing can add significantly to the cost of a dietary supplement. The cheapest supplement is not always the best buy, though a high price does not necessarily guarantee high quality. It is important to investigate the supplement manufacturer whose products are being considered. Manufacturer contact information appears on the supplement facts label. One should inquire whether the manufacturer uses Good Manufacturing Practices, how they ensure the purity of their ingredients, and whether they have their products tested by independent laboratories to verify that the label accurately reflects the product in the supplement container.
Alternative therapies are increasingly being used to complement conventional medicine. The consumer should be knowledgeable of the modality chosen, its intended purpose, and whether it is appropriate for that purpose. In the case of dietary supplements, consumers should educate themselves about the appropriate application for the supplement and the dose that is known to be safe and effective. Although many reputable CAM practitioners and dietary supplement manufacturers exist, consumers should educate themselves about the hallmarks of a quality practitioner or dietary supplement. With CAM modalities in general, and dietary supplements in particular, it is helpful to identify health care professionals who are knowledgeable about CAM and who can provide help in using CAM effectively.
SEE ALSO Dietary Supplements ; Macrobiotic Diet .
Ruth M. DeBusk
Bibliography
Astin, John A. (1998). "Why Patients Use Alternative Medicine: Results of a National Study." Journal of the American Medical Association 279:1548–1553.
Eisenberg, David M.; Davis, Roger B.; Ettner, Susan L.; Appel, Scott; Wilkey, Sonja; Van Rompay, Maria; Kessler, Ronald C. (1998). "Trends in Alternative Medicine Use in the United States, 1990–1997: Results of a Follow-Up National Survey." Journal of the American Medical Association 280:1569–1575.
Eisenberg, David M.; Kessler, Ronald C.; Foster, Cindy; Norlock, Frances E.; Calkins, David R.; and Delbanco, Thomas L. (1993). "Unconventional Medicine in the United States: Prevalence, Costs, and Patterns of Use." New England Journal of Medicine 328:246–252.
Internet Resources
Center for Food Safety and Applied Nutrition. "Dietary Supplements." Available from <http://www.cfsan.fda.gov>
Federation of State Medical Boards. "Member Medical Boards." Available from <http://www.fsmb.org/members.htm>
National Center for Complementary and Alternative Medicine. <http://www.nccam.nih.gov>
Office of Disease Prevention and Health Promotion (1997). "Report of the Commission on Dietary Supplement Labels." Available from <http://web.health.gov/dietsupp>
U.S. Food and Drug Administration (1995). "Dietary Supplement Health and Education Act." Available from <http://www.fda.gov>

Comment about this article, ask questions, or add new information about this topic: