By the start of the 20th century, physicists had worked with Newton's laws so thoroughly that some of them thought that they were coming to the end of physics. In their opinion, not much was left to do to make physics a complete system. Little did they know that the world they described was soon to be understood in a completely different way. The quantum revolution was about to happen.
This revolution was begun by a very unlikely person, a physicist named Max Planck, who was very conservative in all his views. It speaks well of Planck's intellectual honesty that he was able to accept the reality of what he discovered, even though he found the consequences of his discoveries distasteful and unpleasant for the rest of his life.

Born in 1853, Max Planck came from a conservative and respectable family in Kiel, Germany. Young Max was very bright, and had a variety of fields from which to choose to study for his professional life. Planck chose physics because he felt that it was the field in which he was most likely to do some original work. At the young age of 21, he received his doctorate in physics from the University of Munich.
Planck was investigating the properties of heat- and light-emitting bodies. Classical physics had theories which predicted that the brightness of a body increases continuously as the frequency [3] of its electromagnetic radiation [4] is increased.
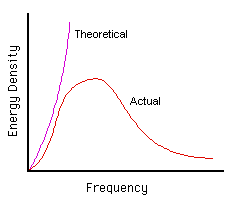
Unfortunately, experiments revealed a totally different picture. The brightness did increase initially, but only upto a limit. Then, actually, it began to fall. We thus get a bell-shaped curve if we plot frequency against brightness.
Besides, another observation was made: as bodies become hotter, their maximum brightness shifts towards higher frequencies. This is why an object, heated to 300-400 C, emits mostly infra-red or heat waves. As the temperature is increased, the object appears to be red, then orange, and finally white or even blue.
Classical theories totally failed to explain this discrepancy between the known facts and the observations. Then, in the winter of 1900, Max Planck found a solution to this problem. Planck ushered in the quantum era by making a bizarre assumption:
Emission and absorption of energy can occur only in discrete amounts.
This might seem totally unsurprising to you, but believe me, it shook the scientists of that period. Planck himself did not know he would end up with this statement!
Imagine for a moment that you are a sculptor, and you have obtained a piece of stone in the shape of a cube. To begin your sculpture, you take a chisel and place its edge against the stone, and then strike the chisel with a hammer. What do you imagine would happen? I think you would imagine that a piece of the stone would be split off, as well as some smaller splinters and pieces of stone. Imagine instead that when you struck the stone, it broke into hundreds of small cubes, each one of them exactly the same size, 3 centimeters per side. Wouldn't you be surprised, even shocked? Imagine furthermore that no matter how hard you tried, these smaller cubes could not be broken into smaller pieces at all!
We think that the reaction that a sculptor would have in such a circumstance would be similar to what Planck and other physicists felt upon discovering that energy only occurred in discrete amounts. It was a completely unexpected discovery, and yet it was only the beginning of what would come later.
Planck called these discrete lumps as quanta. This was against the entire world-view that had been built from the time of Newton onward. In the physics that had been built up since the time of Newton, and indeed in the minds of most thinkers before Newton, matter and energy were thought to be smooth and continuous. Even by the time of Planck, the idea that matter could be ultimately broken down into tiny indivisible 'atoms' was only held by a few physicists.
And so the quantum revolution began.
Footnotes
1. Natural Philosophy was the term used in Isaac Newton's time for what we now call 'the sciences'. 'Science' would only become distinct from philosophy much later. The term 'scientist' was only coined in the middle of the 19th century. Back2. Newton's physics gave a very good understanding of the working of gravity, as well as other natural phenomena. Although Newton's interpretation of his discoveries has mostly been abandoned, the mathematical laws that he discovered are close enough to what is actually found in the real world that they are still useful for many things, from engineering to astrophysics. Back
3. Frequency is a term associated with waves, and refers to the number of waves that pass through a point in a given time interval. For example, if you can count three 'heads' of ripples pass by you in a pond in ten seconds, then the frequency is 3/10 Hz. Hertz is the unit of frequency. Back
4. Electromagnetic radiation consists of fluctuating electric and magnetic fields. In the increasing order of frequency, radio waves, microwaves, infra-red radiation, the visible light (red through violet), ultra-violet radiation, X rays and gamma rays are all electromagnetic waves. They are caused by changes in the electric field or the magnetic field, and they travel at the speed of light, which is 300,000 km per second. Back
* All scientists portraits courtesy: VigyanPrasar.com